Designed for GMP environments. Fully compliant with FDA 21 CFR Part 11, offering end-to-end statistical support from Audit Trails to Process Validation.
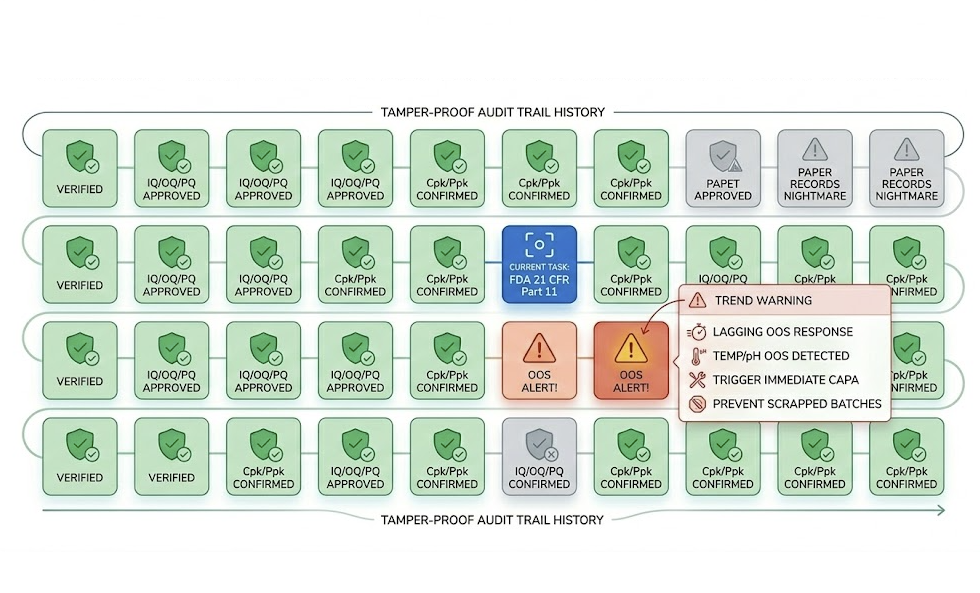
Paper BPRs: Pen errors, lost records, untraceable edits... These are 'fatal wounds' in an audit.
IQ/OQ/PQ stages need massive data to prove Cpk/Ppk. Excel is inefficient, easily modified, and lacks Audit Trails.
Temp/pH OOS requires immediate CAPA. Without real-time alarms, expensive batches are scrapped by the time it's noticed.
Make 'Data Integrity, Validation Evidence, Environmental Monitoring, and Personnel Consistency' a system capability, not a temporary fix.
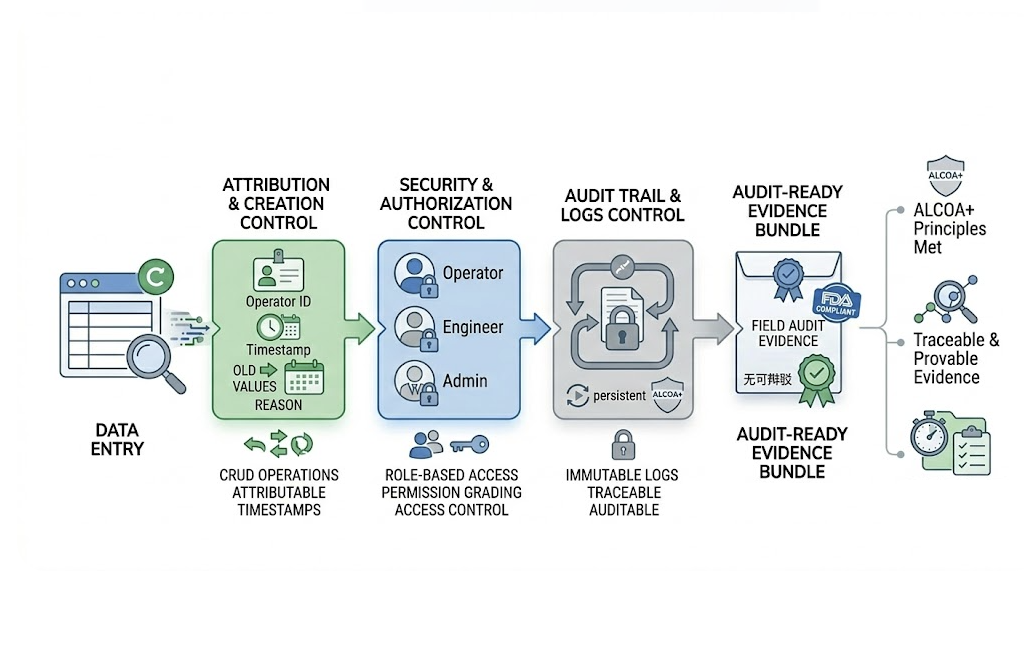
NEXSPC System Kernel · Security & Audit
Make every data entry irrefutable 'audit-ready evidence'. NEXSPC strictly adheres to regulatory requirements at the architectural level.
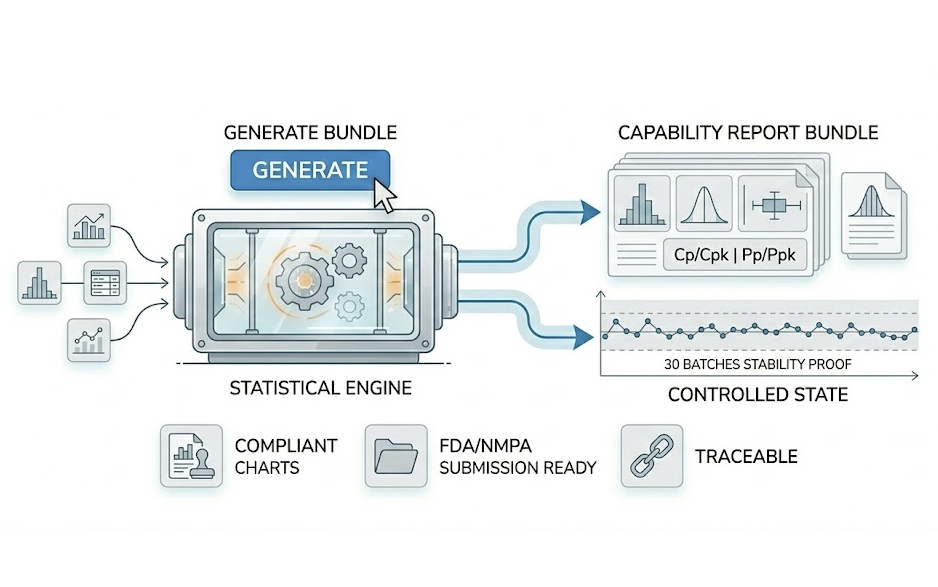
Validation
Prove the process is 'robust and reliable'. In PPQ stages, strong statistical evidence is required for submission and audit.
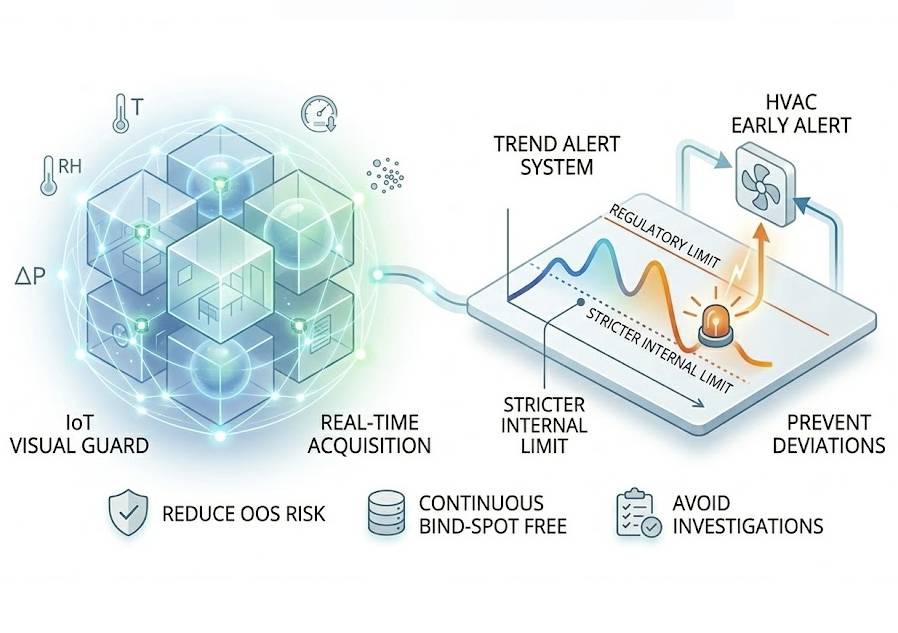
IoT EMS
Ensure every pill is produced in a qualified environment. GMP strictly requires 24/7 blind-spot-free monitoring of Temp/RH, Differential Pressure, and Particles.
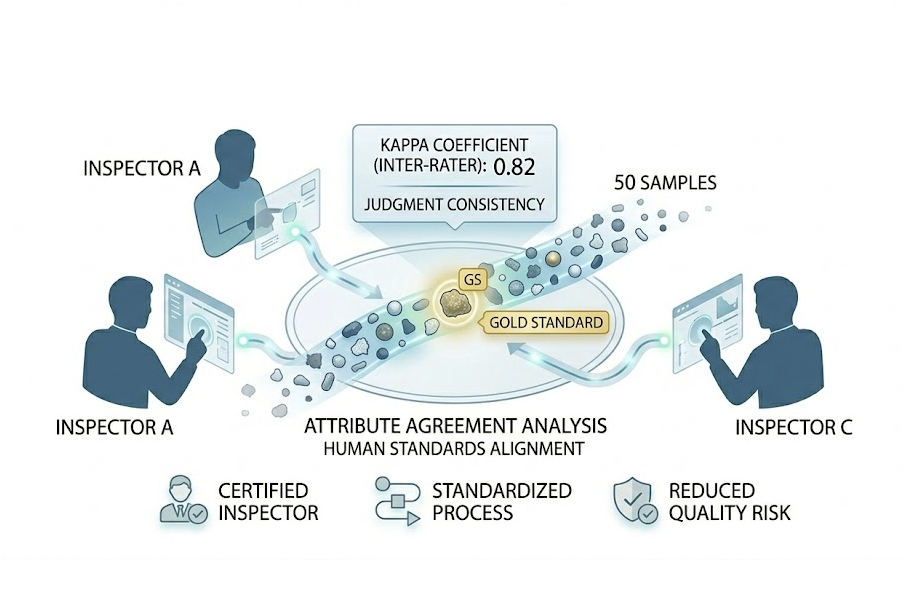
Attribute MSA
Unify human judgment standards to reduce false negatives and false positives. Manual visual inspection (e.g., for particles) relies on humans and needs stats for consistency.
Transform compliance from 'formality' to a 'systematic chain of evidence', ensuring stability in audits and submissions.
| Dimension | Traditional Excel / Paper Mode | NEXSPC Pharma Compliance Ed. |
|---|---|---|
| Data Integrity | Paper tears/lost; Excel modified without trace. | Full Audit Trail, meets ALCOA+ principles. |
| OOS Management | Manual deviation forms after OOS; slow process. | Instant OOS alarm; system auto-logs event timestamp. |
| Validation Efficiency | Weeks of manual data compiling; error-prone. | Auto-aggregation of batch data; one-click validation report. |
| Env. Monitoring | Manual scheduled patrolling; data gaps exist. | Sensors collect continuously 24/7; no blind spots. |
Understood our solutions? Are you ready? Click the button on the right to View Pricing or Consult Now.